|
Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. GBD 2016 Disease and Injury Incidence and Prevalence Collaborators. Junctate boosts phagocytosis by recruiting endoplasmic reticulum Ca 2+ stores near phagosomes. ER-mitochondria microdomains in cardiac ischemia-reperfusion injury: a fresh perspective. Mechanistic insights in recov-ER-phagy: micro-ER-phagy to recover from stress. Quality control in the endoplasmic reticulum: crosstalk between ERAD and UPR pathways. Recent insights into mammalian ER-PM junctions. Endoplasmic reticulum stress signalling and the pathogenesis of non-alcoholic fatty liver disease. Functional diversification of ER stress responses in Arabidopsis. Mechanisms, regulation and functions of the unfolded protein response. Strategies to reduce ER stress (such as small-molecule proteostasis promoters and gene therapy) help to stabilize misfolded proteins and promote correct protein folding, thereby contributing to the prevention and management of CVD. The endoplasmic reticulum (ER) regulates crucial processes governing cardiovascular function ER and mitochondria contacts assist in the transport of mitochondrial Ca 2+, and abnormalities cause cardiomyocyte mitochondrial dysfunction.ĮR stress can be beneficial through an adaptive unfolded protein response (UPR) or detrimental through a maladaptive UPR excessive ER stress perturbs the function of secretory pathways, contributing to cardiovascular pathology.ĮR stress can be either a cause or a consequence of cardiovascular disease (CVD) disrupted ER homeostasis provokes the onset of CVD, which further exacerbates ER stress, creating a vicious cycle.Ĭanonical and non-canonical ER stress signalling can contribute to either cardiovascular protection or pathology, depending on the cellular environment and disease progression status.Ĭlassic ER stress sensors (such as inositol-requiring protein 1α) also have a non-canonical function, such as scaffolding between cellular organelles. We also summarize the latest advances in our understanding of the importance of the UPR and ER stress in the pathogenesis of CVD and discuss potential therapeutic strategies aimed at restoring ER proteostasis in CVDs. In this Review, we discuss the distinct roles of the UPR and ER stress response as both causes and consequences of CVD. 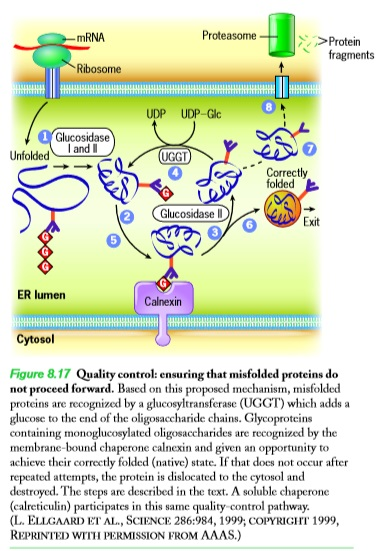
ER stress functions as a double-edged sword, with long-term ER stress resulting in cellular defects causing disturbed cardiovascular function. When the adaptive UPR fails to preserve ER homeostasis, a maladaptive or terminal UPR is engaged, leading to the disruption of ER integrity and to apoptosis. ER proteostasis is governed by the unfolded protein response (UPR), a signalling pathway that adjusts the protein-folding capacity of the cell to sustain the cell’s secretory function. Although specific CVDs and the associated cardiometabolic abnormalities have distinct pathophysiological and clinical manifestations, they often share common traits, including disruption of proteostasis resulting in accumulation of unfolded or misfolded proteins in the endoplasmic reticulum (ER). Cardiovascular diseases (CVDs), such as ischaemic heart disease, cardiomyopathy, atherosclerosis, hypertension, stroke and heart failure, are among the leading causes of morbidity and mortality worldwide.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
